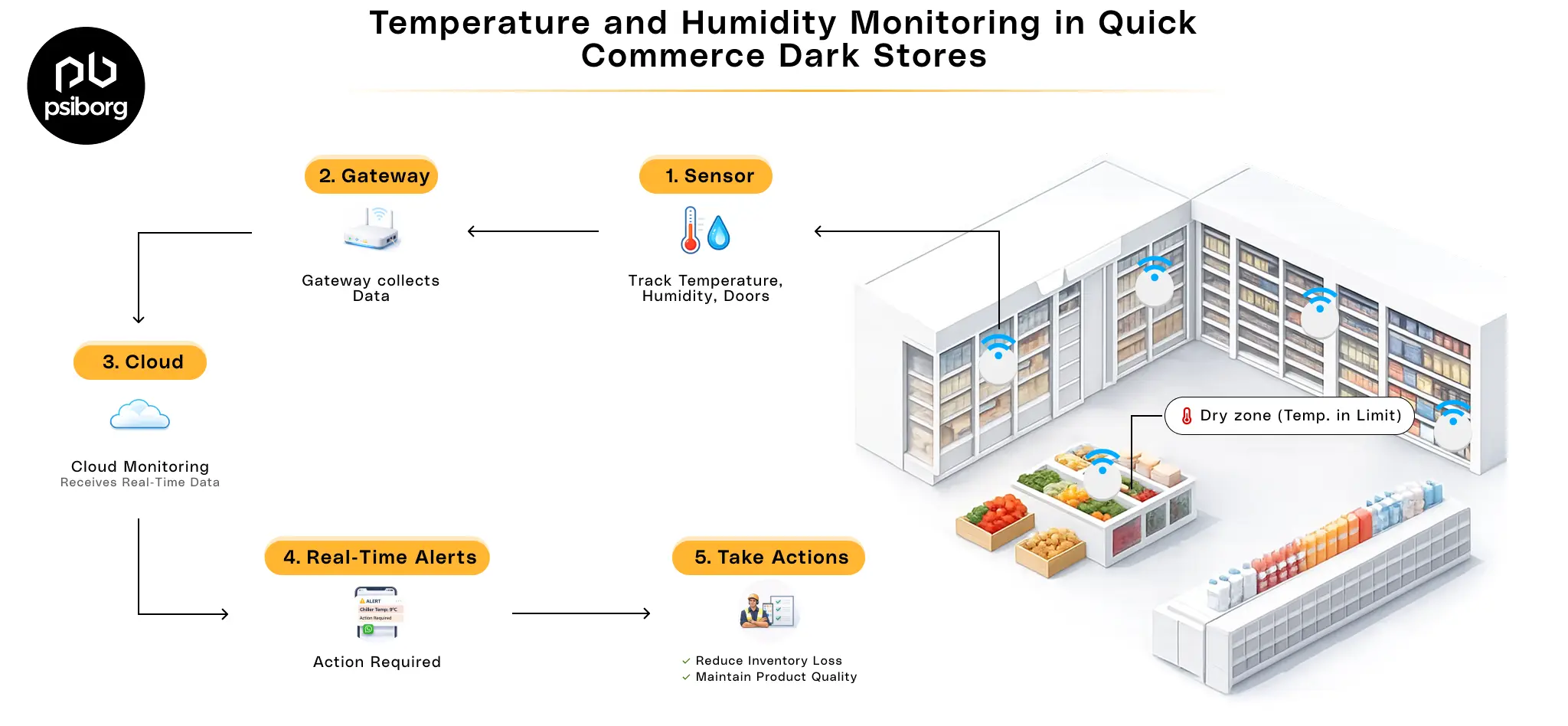
In pharmaceutical manufacturing and storage, even small environmental changes can create big problems. A slight temperature or humidity deviation can compromise drug stability, trigger compliance violations, or result in costly product rejection.
In fact, the pharmaceutical industry loses an estimated USD 35 billion every year due to temperature-related failures across the supply chain.
The impact is even more critical in sensitive products. Studies show temperature excursions contribute to up to 25% loss in vaccine effectiveness, costing the industry billions in waste and compliance penalties.
For businesses, understanding temperature humidity monitoring in pharmaceutical industries isn’t just technical, it’s strategic.
This complete guide walks you through why precise environmental monitoring matters, how IoT solutions mitigate risk, and what best practices keep your products safe, compliant, and profitable.
IoT Solutions for Temperature & Humidity Monitoring In Pharmaceutical Industries
IoT-based temperature and humidity monitoring systems are built to continuously measure, record, and control environmental conditions that directly affect drug quality and compliance. Unlike traditional data loggers, these systems provide real-time visibility and automated response across all critical pharmaceutical environments.
Once the right controls are in place, the next question is how real-time temperature and humidity monitoring actually functions in a regulated pharma environment.
How IoT Temperature & Humidity Monitoring Works?
A pharma-grade monitoring system is designed to deliver validated, continuous, and audit-ready environmental data across all critical zones:
- Calibrated temperature and humidity sensors capture readings at fixed, GMP-defined intervals to maintain stability conditions
- Gateways or edge devices transmit data instantly to a secure central system, eliminating manual data collection
- Centralized monitoring software applies validated limits, timestamps every record, and maintains data integrity
- Automated alerts and escalations notify responsible teams the moment temperature or humidity deviates from approved ranges.
Also read, how our Temperature and Humidity monitoring in the warehouse ensures optimal storage conditions.
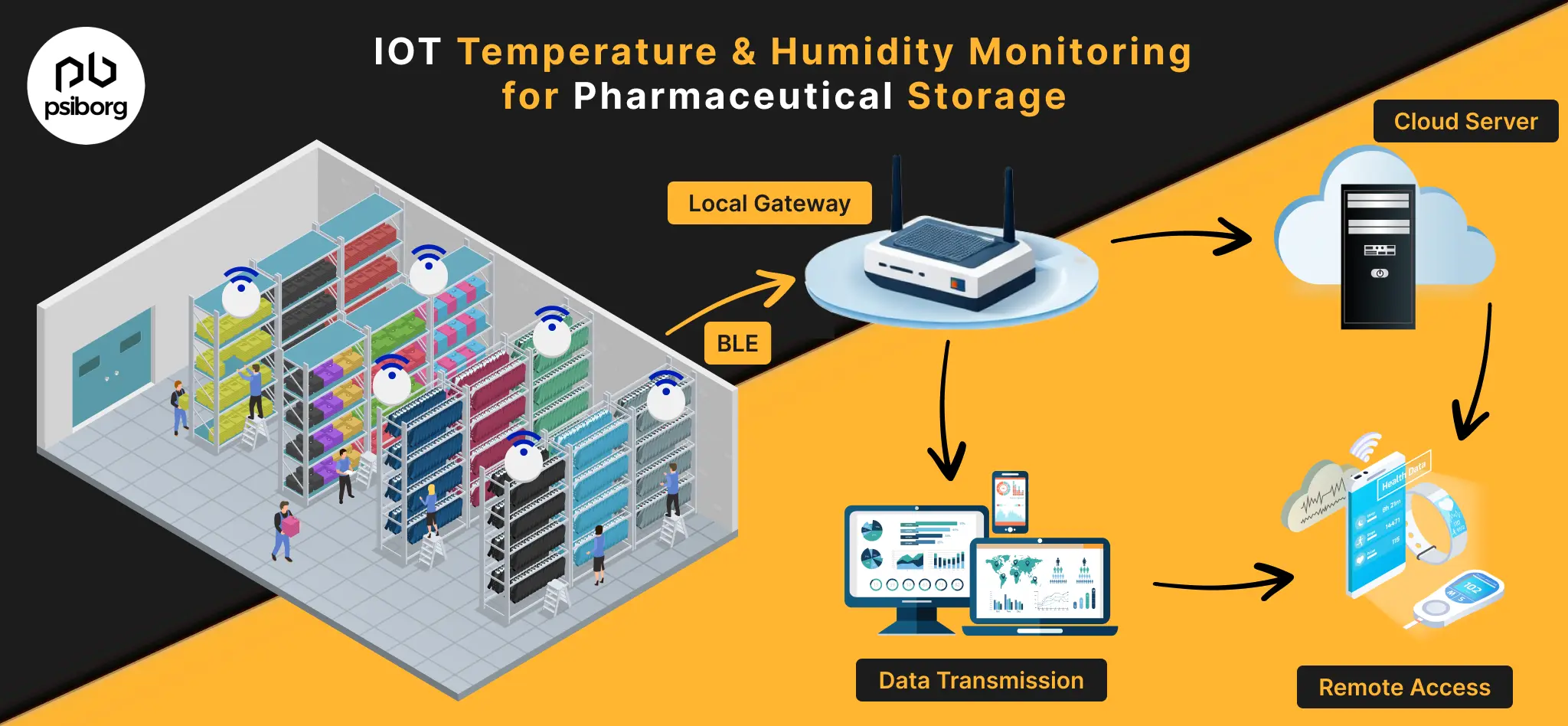
Detailed IoT Architecture for Pharmaceutical Monitoring
An IoT architecture for pharmaceutical temperature and humidity monitoring is designed to deliver continuous visibility, data integrity, and audit readiness across all controlled zones.
- Sensor layer: Validated, calibrated temperature and humidity sensors are placed at critical control points based on heat-mapping and risk analysis to capture accurate, GMP-compliant data.
- Edge and gateway layer; Gateways securely collect and transmit readings in real time, buffer data during network outages, and apply encryption to maintain continuity and security.
- Monitoring platform layer: Central software enforces approved thresholds, triggers instant alerts on excursions, logs deviations, and maintains tamper-proof, time-stamped audit trails.
- Data storage and integration layer: Compliance-ready storage supports long-term retention, role-based access, and integration with QMS and CAPA systems for inspection-ready reporting.
Even the best tech stack falls short if the system lacks features that directly support compliance, risk reduction, and operational efficiency in pharma settings.
Why Temperature & Humidity Monitoring Matters in Pharma
Temperature and humidity directly influence drug stability, safety, and regulatory compliance across the pharmaceutical lifecycle. Unlike many industries, pharma products are chemically and biologically sensitive, meaning even short-term environmental deviations can permanently damage product integrity.
1. Direct Impact on Drug Stability and Potency
Most pharmaceutical products are developed and approved based on strict stability conditions. When temperature or humidity moves outside validated limits:
- Active Pharmaceutical Ingredients (APIs) can degrade faster, reducing potency.
- Moisture exposure can cause tablet swelling, cracking, or dissolution failure.
- Biologics and vaccines may suffer irreversible loss of efficacy, even if conditions normalize later.
This is why cold chain monitoring pharmaceutical operations is tightly regulated; once a breach occurs, products often cannot be requalified.
2. Compliance Risk Extends Beyond Manufacturing
Regulatory expectations apply across the entire product lifecycle, including:
- Manufacturing and cleanrooms
- Warehouses and cold storage
- Stability chambers
- Transportation and last-mile delivery
A single unmonitored zone can trigger audit observations, batch rejection, recalls, or delayed releases. A centralized Pharma environmental monitoring system ensures continuous tracking, secure data logging, and audit readiness across all areas.
3. Small Deviations, Large Financial Losses
Environmental failures are often silent. Just a few hours of exposure can result in:
- Entire batch write-offs
- Re-manufacturing costs
- Missed delivery commitments
- Revenue and reputational loss
Without real-time alerts and escalation, issues are typically discovered after damage is irreversible.
Also check, how our remote grain moisture monitor maintains grain quality.
4. From Compliance Burden to Operational Advantage
Modern pharma companies are moving beyond manual logs to intelligent monitoring. A connected monitoring system enables:
- Immediate deviation detection
- Faster corrective action
- Predictable compliance outcomes
- Lower quality and operational risk
In short, temperature and humidity monitoring is no longer just about meeting regulations; it’s about protecting revenue, accelerating audits, and maintaining trust in an increasingly regulated market.
Regulatory Requirements for Temperature & Humidity Monitoring in Pharmaceutical Industries
Pharmaceutical regulators don’t just expect controlled environments; they expect proof of continuous control. Temperature and humidity monitoring is embedded into global GMP and GDP frameworks because environmental deviations directly affect product quality, patient safety, and data integrity.
Pharma companies typically operate under multiple regulatory bodies, each with aligned expectations:
- WHO GMP: Requires continuous monitoring of storage and manufacturing environments, with documented evidence of control and deviation handling.
- US FDA (21 CFR Part 210 & 211): Mandates suitable environmental conditions, calibrated instruments, and complete records to demonstrate product integrity.
- EU GMP (Annex 1 & Annex 15): Emphasizes environmental monitoring, validation, and documented control of critical parameters.
- ICH Q1A (Stability Testing Guidelines): Defines approved temperature and humidity conditions used to establish product shelf life.
Here are the regulated temperature & humidity conditions by area:
Area | Typical Requirement |
Manufacturing & Cleanrooms | Controlled, validated ranges per process |
Warehouses | Ambient or controlled storage with continuous logs |
Cold Rooms | 2–8 °C with alarms and excursion handling |
Stability Chambers | ICH-defined temperature & RH conditions |
Transport & Distribution | Continuous monitoring with traceability |
Once regulatory expectations are clear, the real challenge begins, maintaining continuous control across complex pharma environments without gaps, delays, or data blind spots.
Key Challenges in Pharmaceutical Temperature & Humidity Monitoring
Despite strict regulations, many pharma companies struggle to maintain consistent environmental control. These challenges are rarely about intent; they stem from operational complexity in Pharma Lifecycle.
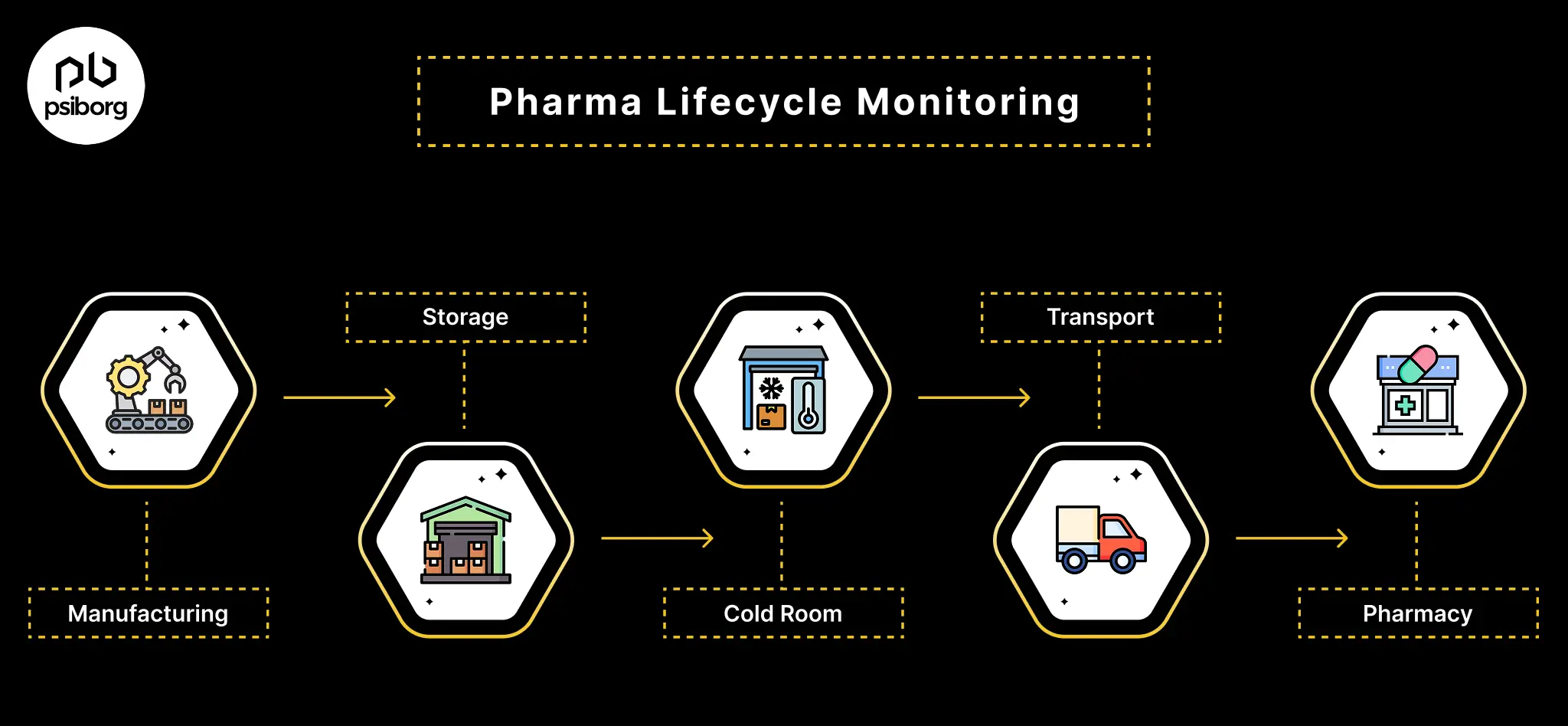
- Delayed detection of excursions: Manual or periodic pharmaceutical temperature monitoring identifies deviations hours or days late, leaving no time for corrective action and forcing reactive investigations.
- Limited visibility across operations: Monitoring gaps across cleanrooms, warehouses, cold rooms, stability chambers, and in-transit storage prevent lifecycle-wide risk visibility and weaken compliance.
- Fragmented and siloed systems: Disconnected tools create data blind spots, increasing audit exposure and complicating root-cause analysis.
- Manual processes and human error: Paper logs and spreadsheets lead to missed readings, inconsistent records, and data gaps that auditors quickly flag, especially when IoT temperature monitoring for pharmaceuticals is an accepted standard.
- No automated alerts or escalation: Systems that only record data fail to trigger immediate alerts, escalation workflows, or CAPA actions when limits are breached.
- Data integrity and audit readiness gaps: Editable records, missing time stamps, and incomplete audit trails undermine inspection readiness and regulatory confidence.
To overcome delayed detection, data gaps, and audit risk, pharmaceutical companies are increasingly adopting purpose-built systems designed specifically for continuous temperature and humidity control.
Features in a Pharma-Grade Temperature & Humidity Monitoring System
When evaluating a temperature and humidity monitoring solution for pharmaceutical operations, businesses should look beyond basic data logging and focus on features that actively protect product quality and compliance.
- Real-time monitoring and alerts: Continuous data capture with instant alerts via SMS, email, or dashboard ensures deviations are addressed before stability is compromised.
- Validated thresholds and zoning; Configurable limits by room, product, or storage type support GMP requirements and prevent false compliance across mixed-use areas.
- Audit-ready data integrity: Time-stamped, tamper-proof records with role-based access and electronic signatures simplify inspections and meet regulatory expectations.
- Calibration and sensor health tracking; Automated reminders and calibration history ensure sensors remain accurate and inspection-ready.
- Deviation management and reporting: Built-in deviation logs, trend analysis, and exportable reports support investigations, CAPA, and regulatory submissions.
- Scalability and system integration: The ability to scale across sites and integrate with QMS, ERP, or cold chain systems reduces manual effort and long-term costs.
Final Thoughts
Temperature and humidity control in pharmaceutical environments is no longer just about meeting regulatory checklists. It directly impacts product stability, compliance outcomes, audit timelines, and financial risk. As regulations tighten and supply chains become more complex, manual or fragmented monitoring systems leave too much room for error.
PsiBorg acts as a full-stack IoT service partner, delivering integrated hardware and software tailored to pharmaceutical operations. From validated temperature and humidity sensors and secure gateways to real-time monitoring platforms, alerts, audit-ready data, and system integrations, PsiBorg designs solutions that fit your exact compliance and operational needs across manufacturing, storage, and cold chain environments.
Partner with PsiBorg to deploy a customized IoT temperature & humidity monitoring solution built for pharmaceutical excellence.
FAQs
Why is temperature and humidity monitoring critical in the pharmaceutical industry?
Because even minor deviations can degrade APIs, reduce drug potency, and cause batch failures. Continuous monitoring protects product quality and prevents costly recalls. It also ensures ongoing GMP and GDP compliance.
What are the GMP and GDP requirements for temperature monitoring in pharmaceuticals?
GMP and GDP require continuous monitoring, documented evidence, alarm management, and audit-ready records. Data must be accurate, traceable, and tamper-proof. Manual logs alone are no longer sufficient.
How does humidity affect drug stability and shelf life?
Excess humidity can cause tablet swelling, cracking, microbial growth, and chemical degradation. Moisture-sensitive formulations lose stability faster. Controlled humidity directly extends shelf life and efficacy.
What is the best temperature and humidity monitoring system for pharmaceutical storage?
A system with calibrated sensors, real-time alerts, centralized dashboards, and compliance-ready data storage. IoT-based systems outperform standalone data loggers. Customization for zones and products is key.
How does an IoT-based temperature monitoring system help pharmaceutical companies?
It detects deviations in real time, automates alerts, and maintains audit-ready records. This reduces product loss, speeds corrective action, and simplifies inspections. It turns compliance into a proactive process.
How often should temperature and humidity be recorded in pharma warehouses and cold rooms?
Regulations expect continuous monitoring, not periodic checks. Data is typically captured every few minutes with real-time alerts. Frequency depends on risk level, product sensitivity, and regulatory guidelines.